- Home
- About DGR
- How to use facility
- Facilities
- Elemental Analyzer (CHNS/O)
- Total Organic Caron Analyzer (TOC)
- High Performance Liquid Chromatography
- Microplate Photometer (ELISA reader)
- Ion Chromatograph
- Nitrogen Analyser
- Near Infra-red (NIR) Analyser
- Atomic Absorption Spectrometer
- Flow Cytometer
- Karyotype Analyser
- GenomeLab GeXP Genetic Analysis System
- Automated Pipetting System
- Real time PCR
- Cryostat Microtome Automatic (Deluxe)
- Contact Us
- Usefull Links

| ||||||||||||||||||||||||||||||||||||||
Atomic Absorption Spectrometer
| ||||||||||||||||||||||||||||||||||||||
Instrument Details
 Make : PerkinElmer Model : AAnalyst 400 with HGA 900 Specification : This Flame/furnace Atomic absorption spectrophotometer (AAS) easily determine low concentration of various elements (Cu, Fe, Mn, Zn, K, Na, Mg, Mo, Co, Ni, Ca, Pb, Cd, Al, Cr, Si, Ag) through flame atomic absorption (AA) as well a few (K and Na) in emission mode in the solution. 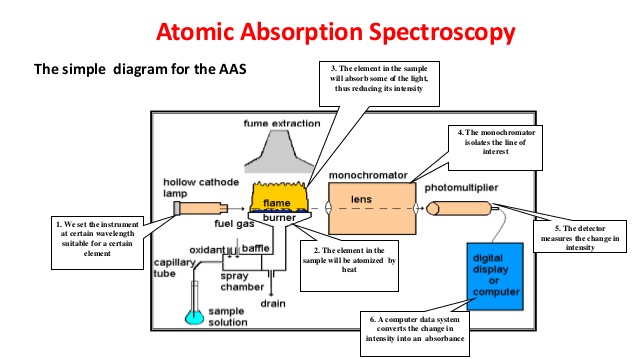 Graphite Furnace HGA 900
MAIN FEATURES OF THE INSTRUMENT
1. Double beam optics, autosampler with high efficient segmented solid state detector 2. Operates under Emission and Absorption modes 3. Graphite furnace HGA 900 for ultralow detection of elements | ||||||||||||||||||||||||||||||||||||||
|
Applications
• Agriculture • Environmental • Food analysis • Geochemical/Mining Industry • Petrochemical Industry |
References
1. B. Welz, M. Sperling (1999), Atomic Absorption Spectrometry, Wiley-VCH, Weinheim, Germany, ISBN 3-527-28571-7.
|
User Instructions
1. Only plant, soil, water and fertilizer samples. 2. Sample of minimum 20 ml for liquid and 2 g for solid samples |
Contact Us
Contact : 0285 2673041
Email Id : amrit.singh@icar.gov.in
| |||||||||||||||||||||||||||||||||||
Atomic Absorption Spectrometer charges (Rs)includes GST
| ||||||||||||||||||||||||||||||||||||||

